The distribution of singly ionized ionospheric helium from 304 A backscatter observations
Important lines are the hydrogen Balmer lines, lines of neutral and singly ionized helium, iron lines, the H and K doublet of ionized calcium at 396.8 and 393.3 nm, the G band due to the CH molecule, the 422.7 nm neutral calcium line, several metal lines around 431 nm, and the lines of titanium oxide. Question: What Wavelength Corresponds To The N = 2 - N = 1 Transition In Singly-ionized Helium? A) 11.4 Nm B) 30.4 Nm C) 22.8 Nm D) 4.56 Nm E) 15.2 Nm. This problem has been solved! Oh darling beatles guitar pro. SINGLY IONIZED HELIUM ATOM Singly ionized helium is an atom that has lost one of its electrons. We must expect its remaining electron to act like a hydrogen electron. There is only one difference between a hydrogen atom and a singly ionized helium atom; number of protons in the nucleus, so the nuclear charg.
Abstract
The theoretical distribution of helium ions in the ionosphere is studied as a function of such ionospheric parameters as the remaining ion composition and distribution, temperature, magnetic field topology, and ionospheric dynamics. An attempt is made to verify the theoretical predictions of the H(+) distribution in the ionosphere on the basis of observations of 304 A radiation resonantly scattered from He(+).
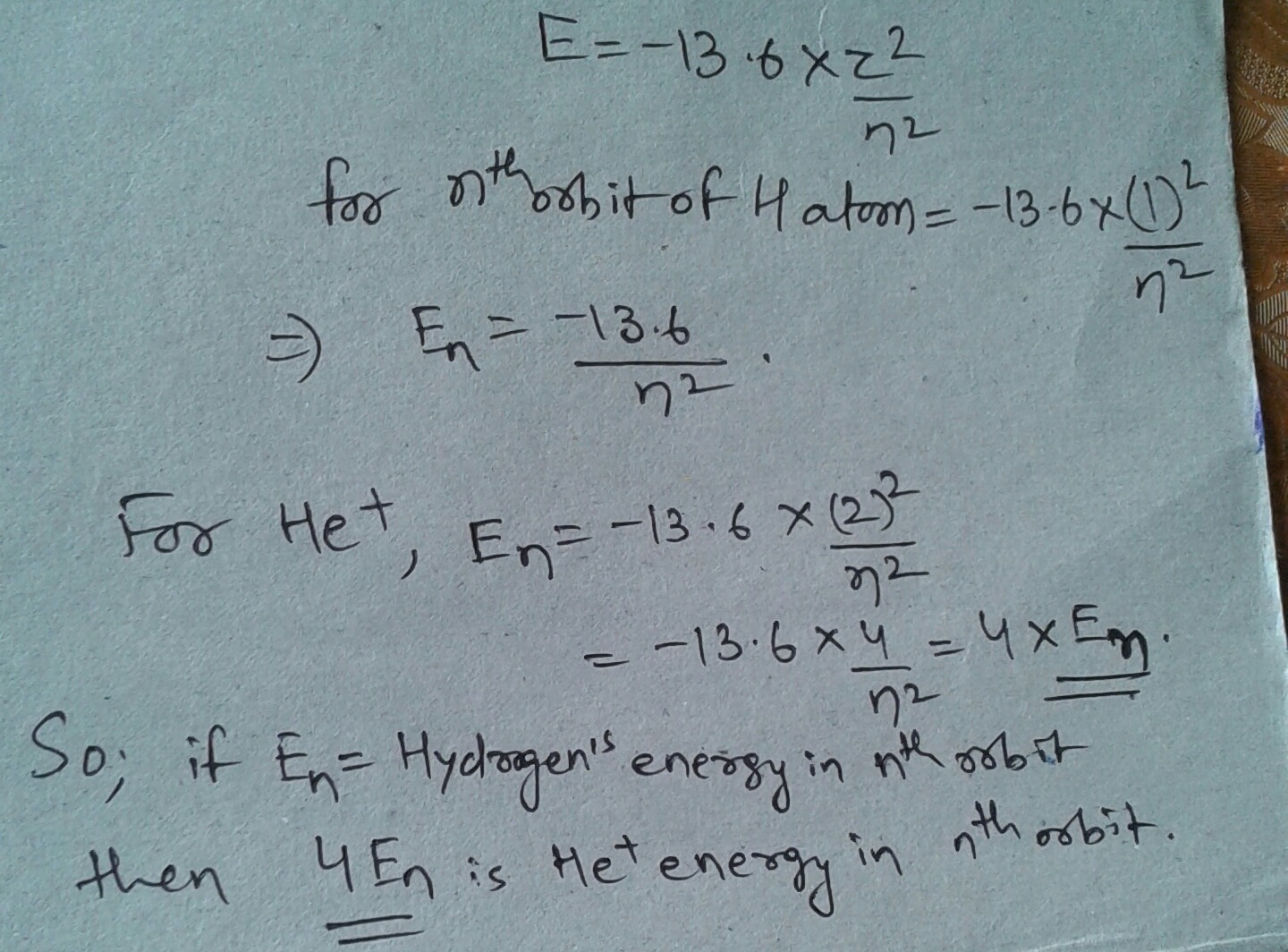
Ionized Helium
Singly Ionized Helium
- Atmospheric Scattering;
- Helium Ions;
- Ionospheric Ion Density;
- Atmospheric Composition;
- Atmospheric Temperature;
- Backscattering;
- Magnetic Fields;
- Rocket Sounding;
- Topology;
- Geophysics
