Teletype for Atom
Great things happen when developers work together—from teaching and sharing knowledge to building better software. Teletype for Atom makes collaborating on code just as easy as it is to code alone, right from your editor.
Share your workspace and edit code together in real time. To start collaborating, open Teletype in Atom and install the package. Neighborhood online garage sale.
GitHub for Atom
A text editor is at the core of a developer’s toolbox, but it doesn't usually work alone. Work with Git and GitHub directly from Atom with the GitHub package.
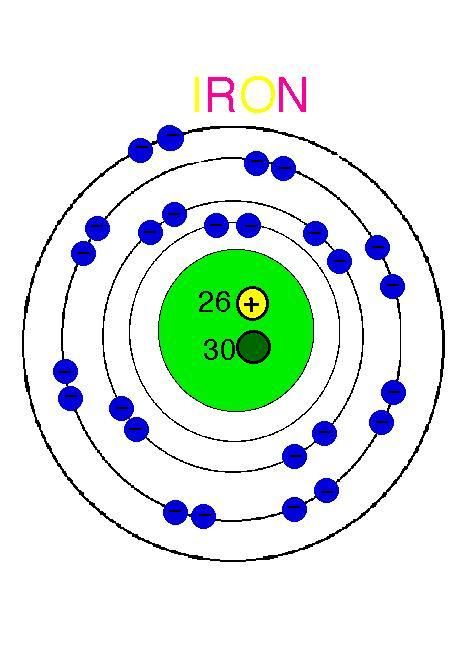
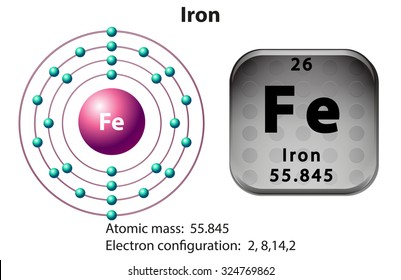
Basic Information. Atomic Number:26. Atomic Mass:55.845 amu. Melting Point:1535.0 °C (1808.15 K, 2795.0 °F) Boiling Point:2750.0 °C (3023.15 K, 4982.0 °F) Number of Protons/Electrons:26. Number of Neutrons:30. The iron atom, which is given the symbol Fe, has an atomic weight of 56 which compares with aluminium, Al, at 27, lead, Pb, at 207 and carbon, C, at 12. In iron at room temperature the atoms are arranged in a regular pattern, or lattice, which is called body centred cubic or bcc for short. The smallest repeatable three dimensional pattern is then a cube with an atom at each corner plus one in the middle of the cube.
Create new branches, stage and commit, push and pull, resolve merge conflicts, view pull requests and more—all from within your editor. The GitHub package is already bundled with Atom, so you're ready to go!
Everything you would expect
Cross-platform editing
Atom works across operating systems. Use it on OS X, Windows, or Linux.
Built-in package manager
Search for and install new packages or create your own right from Atom.
Smart autocompletion
Atom helps you write code faster with a smart and flexible autocomplete.
File system browser
Fe Atomic Name
Easily browse and open a single file, a whole project, or multiple projects in one window.
Multiple panes
Split your Atom interface into multiple panes to compare and edit code across files.
Find and replace
Find, preview, and replace text as you type in a file or across all your projects.
Make it your editor
Packages
Choose from thousands of open source packages that add new features and functionality to Atom, or build a package from scratch and publish it for everyone else to use.
Themes
Atom comes pre-installed with four UI and eight syntax themes in both dark and light colors. Can't find what you're looking for? Install themes created by the Atom community or create your own.
Customization
It's easy to customize and style Atom. Tweak the look and feel of your UI with CSS/Less, and add major features with HTML and JavaScript.
See how to set up Atom
Under the hood
Atom is a desktop application built with HTML, JavaScript, CSS, and Node.js integration. It runs on Electron, a framework for building cross platform apps using web technologies.
Open source
Atom is open source. Be part of the Atom community or help improve your favorite text editor.
Keep in touch
| GitHub | github.com/atom |
| @AtomEditor | |
| Chat | Slack |
| Forum | Discuss |
| Stuff | Atom Gear |
| RSS Feed | Packages & Themes |
Article
Please select which sections you would like to print:
While every effort has been made to follow citation style rules, there may be some discrepancies. Please refer to the appropriate style manual or other sources if you have any questions.
Fe Atomic Structure
Our editors will review what you’ve submitted and determine whether to revise the article.
Join Britannica's Publishing Partner Program and our community of experts to gain a global audience for your work! Articles from Britannica Encyclopedias for elementary and high school students.
The Editors of Encyclopaedia BritannicaEncyclopaedia Britannica's editors oversee subject areas in which they have extensive knowledge, whether from years of experience gained by working on that content or via study for an advanced degree..
Alternative Titles: Fe, ferrum
Iron (Fe), chemical element, metal of Group 8 (VIIIb) of the periodic table, the most-used and cheapest metal.
118 Names and Symbols of the Periodic Table Quiz
The periodic table is made up of 118 elements. How well do you know their symbols? In this quiz you’ll be shown all 118 chemical symbols, and you’ll need to choose the name of the chemical element that each one represents.
| atomic number | 26 |
|---|---|
| atomic weight | 55.847 |
| melting point | 1,538 °C (2,800 °F) |
| boiling point | 3,000 °C (5,432 °F) |
| specific gravity | 7.86 (20 °C) |
| oxidation states | +2, +3, +4, +6 |
| electron configuration | [Ar]3d64s2 |
Occurrence, uses, and properties
Iron makes up 5 percent of Earth’s crust and is second in abundance to aluminum among the metals and fourth in abundance behind oxygen, silicon, and aluminum among the elements. Iron, which is the chief constituent of Earth’s core, is the most abundant element in Earth as a whole (about 35 percent) and is relatively plentiful in the Sun and other stars. In the crust the free metal is rare, occurring as terrestrial iron (alloyed with 2–3 percent nickel) in basaltic rocks in Greenland and carbonaceoussediments in the United States (Missouri) and as a low-nickel meteoric iron (5–7 percent nickel), kamacite. Nickel-iron, a native alloy, occurs in terrestrial deposits (21–64 percent iron, 77–34 percent nickel) and in meteorites as taenite (62–75 percent iron, 37–24 percent nickel). (For mineralogical properties of native iron and nickel-iron, seenative elements [table].) Meteorites are classified as iron, iron-stone, or stony according to the relative proportion of their iron and silicate-mineral content. Iron is also found combined with other elements in hundreds of minerals; of greatest importance as iron ore are hematite (ferric oxide, Fe2O3), magnetite (triiron tetroxide, Fe3O4), limonite (hydrated ferric oxide hydroxide, FeO(OH)∙nH2O), and siderite (ferrous carbonate, FeCO3). Igneous rocks average about 5 percent iron content. The metal is extracted by smelting with carbon (coke) and limestone. (For specific information on the mining and production of iron, seeiron processing.)
| country | mine production 2006 (metric tons)* | % of world mine production | demonstrated reserves 2006 (metric tons)*, ** | % of world demonstrated reserves |
|---|---|---|---|---|
| *Estimated. | ||||
| **Iron content. | ||||
| ***Detail does not add to total given because of rounding. | ||||
| Source: U.S. Department of the Interior, Mineral Commodity Summaries 2007. | ||||
| China | 520,000,000 | 30.8 | 15,000,000,000 | 8.3 |
| Brazil | 300,000,000 | 17.8 | 41,000,000,000 | 22.8 |
| Australia | 270,000,000 | 16.0 | 25,000,000,000 | 13.9 |
| India | 150,000,000 | 8.9 | 6,200,000,000 | 3.4 |
| Russia | 105,000,000 | 6.2 | 31,000,000,000 | 17.2 |
| Ukraine | 73,000,000 | 4.3 | 20,000,000,000 | 11.1 |
| United States | 54,000,000 | 3.2 | 4,600,000,000 | 2.6 |
| South Africa | 40,000,000 | 2.4 | 1,500,000,000 | 0.8 |
| Canada | 33,000,000 | 2.0 | 2,500,000,000 | 1.4 |
| Sweden | 24,000,000 | 1.4 | 5,000,000,000 | 2.8 |
| Iran | 20,000,000 | 1.2 | 1,500,000,000 | 0.8 |
| Venezuela | 20,000,000 | 1.2 | 3,600,000,000 | 2.0 |
| Kazakhstan | 15,000,000 | 0.9 | 7,400,000,000 | 4.1 |
| Mauritania | 11,000,000 | 0.7 | 1,000,000,000 | 0.6 |
| Mexico | 13,000,000 | 0.8 | 900,000,000 | 0.5 |
| other countries | 43,000,000 | 2.5 | 17,000,000,000 | 9.4 |
| world total | 1,690,000,000 | 100*** | 180,000,000,000 | 100*** |
The average quantity of iron in the human body is about 4.5 grams (about 0.004 percent), of which approximately 65 percent is in the form of hemoglobin, which transports molecular oxygen from the lungs throughout the body; 1 percent in the various enzymes that control intracellular oxidation; and most of the rest stored in the body (liver, spleen, bone marrow) for future conversion to hemoglobin. Red meat, egg yolk, carrots, fruit, whole wheat, and green vegetables contribute most of the 10–20 milligrams of iron required each day by the average adult. For the treatment of hypochromic anemias (caused by iron deficiency), any of a large number of organic or inorganic iron (usually ferrous) compounds are used.
Iron, as commonly available, nearly always contains small amounts of carbon, which are picked up from the coke during smelting. These modify its properties, from hard and brittle cast irons containing up to 4 percent carbon to more malleable low-carbon steels containing less than 0.1 percent carbon.
Wolframite Fewo4 Atoms Fe
Get a Britannica Premium subscription and gain access to exclusive content. Subscribe Now
Three true allotropes of iron in its pure form occur. Delta iron, characterized by a body-centred cubic crystal structure, is stable above a temperature of 1,390 °C (2,534 °F). Below this temperature there is a transition to gamma iron, which has a face-centred cubic (or cubic close-packed) structure and is paramagnetic (capable of being only weakly magnetized and only as long as the magnetizing field is present); its ability to form solid solutions with carbon is important in steelmaking. At 910 °C (1,670 °F) there is a transition to paramagnetic alpha iron, which is also body-centred cubic in structure. Below 773 °C (1,423 °F), alpha iron becomes ferromagnetic (i.e., capable of being permanently magnetized), indicating a change in electronic structure but no change in crystal structure. Above 773 °C (its Curie point), it loses its ferromagnetism altogether. Alpha iron is a soft, ductile, lustrous, gray-white metal of high tensile strength.
Pure iron is quite reactive. In a very finely divided state metallic iron is pyrophoric (i.e., it ignites spontaneously). It combines vigorously with chlorine on mild heating and also with a variety of other nonmetals, including all of the halogens, sulfur, phosphorus, boron, carbon, and silicon (the carbide and silicide phases play major roles in the technical metallurgy of iron). Metallic iron dissolves readily in dilute mineral acids. With nonoxidizing acids and in the absence of air, iron in the +2 oxidation state is obtained. With air present or when warm dilute nitric acid is used, some of the iron goes into solution as the Fe3+ ion. Very strongly oxidizing mediums—for example, concentrated nitric acid or acids containing dichromate—passivate iron (i.e., cause it to lose its normal chemical activity), however, much as they do chromium. Air-free water and dilute air-free hydroxides have little effect on the metal, but it is attacked by hot concentrated sodium hydroxide.
Natural iron is a mixture of four stable isotopes: iron-56 (91.66 percent), iron-54 (5.82 percent), iron-57 (2.19 percent), and iron-58 (0.33 percent).
Iron compounds are amenable to study by taking advantage of a phenomenon known as the Mössbauer effect (the phenomenon of a gamma ray being absorbed and reradiated by a nucleus without recoil). Although the Mössbauer effect has been observed for about one-third of the elements, it is particularly for iron (and to a lesser extent tin) that the effect has been a major research tool for the chemist. In the case of iron the effect depends on the fact that the nucleus of iron-57 can be excited to a high energy state by the absorption of gamma radiation of very sharply defined frequency that is influenced by the oxidation state, electron configuration, and chemical environment of the iron atom and can thus be used as a probe of its chemical behaviour. The marked Mössbauer effect of iron-57 has been used in studying magnetism and hemoglobin derivatives and for making a very precise nuclear clock.
Fe Atomic Number
Quick Facts
Fe Atomic Mass
- key people
- related topics
